Delta H in chemistry refers to the change in enthalpy during a chemical or physical process. It represents the amount of heat absorbed or released by a system at constant pressure and is commonly used to describe whether a reaction is exothermic or endothermic.
Chemistry is full of symbols that look intimidating at first but become surprisingly logical once you understand them. One of the most important and frequently used symbols is Delta H. You will see it in thermochemistry, reaction equations, energy diagrams, and even exam questions. Because of this, many students and curious learners ask the same question: what does Delta H mean in chemistry?
In this guide, you will learn what Delta H really represents, why it matters, how it is calculated, and how scientists use it to understand chemical reactions. By the end, Delta H will feel less like a confusing symbol and more like a helpful tool.
What Does Delta H Mean in Chemistry?
In chemistry, Delta H (ΔH) means the change in enthalpy of a system during a process.
The symbol comes from:
- Delta (Δ) meaning “change”
- H representing enthalpy, which is a measure of heat energy
Put simply, Delta H tells us how much heat energy is gained or lost during a reaction at constant pressure.
If heat is released, Delta H is negative.
If heat is absorbed, Delta H is positive.
Understanding Enthalpy Before Delta H
To fully understand Delta H, it helps to know what enthalpy itself means.
Enthalpy is a measure of the total heat content of a system. It includes:
- Internal energy of the system
- Energy required to make room for the system by pushing against surrounding pressure
In everyday chemistry, enthalpy is useful because most reactions occur at constant atmospheric pressure.
As a result, Delta H becomes a practical way to measure heat changes during reactions.
Why Delta H Is Important in Chemistry
Delta H is important because it helps chemists understand how energy behaves in reactions.
Specifically, Delta H allows scientists to:
- Predict whether a reaction releases or absorbs heat
- Compare energy changes between reactions
- Design industrial and laboratory processes
- Understand reaction feasibility and efficiency
Without Delta H, it would be much harder to describe energy changes in a consistent way.
Positive and Negative Delta H Values Explained
One of the first things students notice is that Delta H can be either positive or negative. Each case has a clear meaning.
Positive Delta H: Endothermic Reactions
When Delta H is positive, the reaction absorbs heat from its surroundings.
This means the products have more enthalpy than the reactants.
Examples of endothermic processes include:
- Melting ice
- Evaporation of water
- Photosynthesis
Example equation:
ΔH = +120 kJ
This tells us that 120 kilojoules of heat are absorbed.
Negative Delta H: Exothermic Reactions
When Delta H is negative, the reaction releases heat to the surroundings.
In this case, the products have lower enthalpy than the reactants.
Examples of exothermic reactions include:
- Combustion of fuels
- Neutralization reactions
- Respiration
Example equation:
ΔH = −250 kJ
This means 250 kilojoules of heat are released.
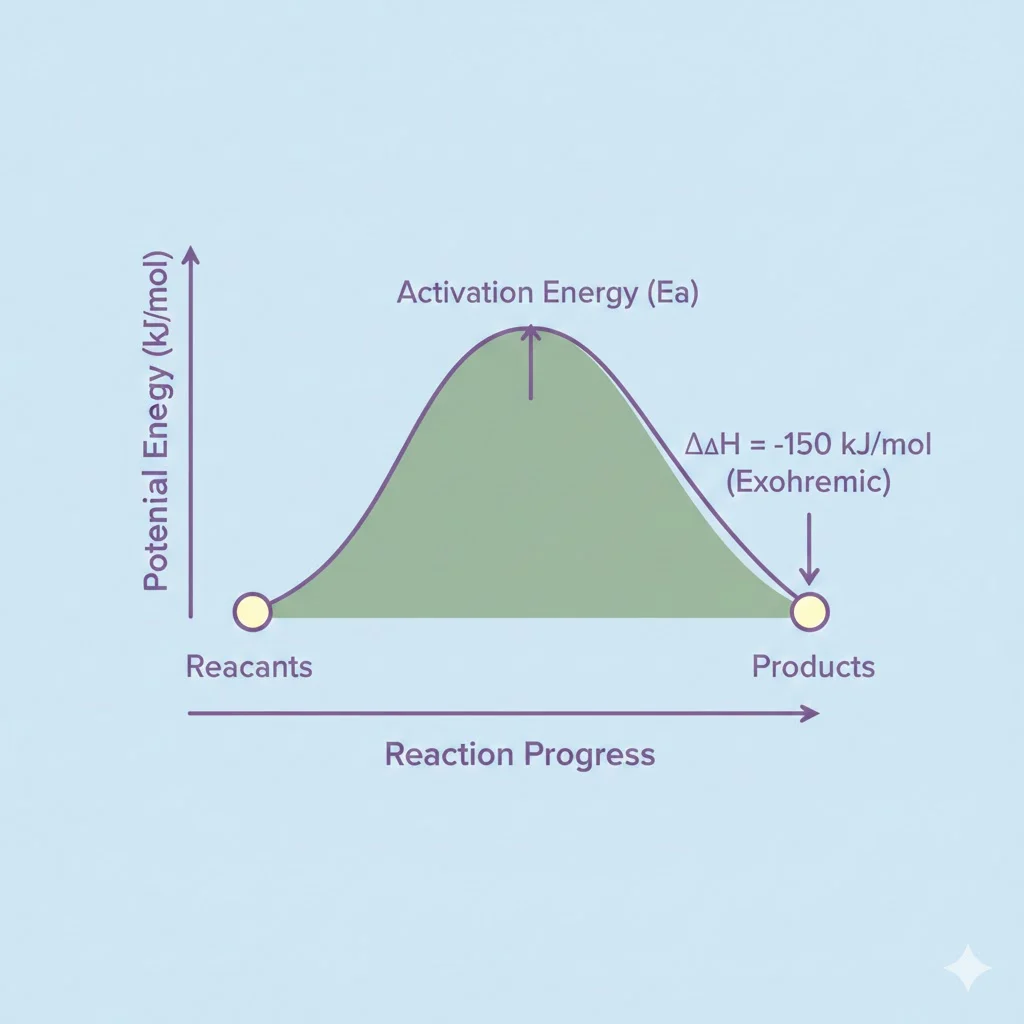
Delta H and Reaction Energy Diagrams
Energy diagrams are often used to visualize Delta H.
In these diagrams:
- The vertical axis represents energy or enthalpy
- The horizontal axis represents reaction progress
If products are lower than reactants, Delta H is negative.
If products are higher than reactants, Delta H is positive.
Because of this, Delta H is simply the vertical difference between reactants and products on the diagram.
How Delta H Is Calculated
Delta H is calculated using the formula:
ΔH = H products − H reactants
This equation shows that Delta H depends on the difference in enthalpy between products and reactants.
If products have lower enthalpy, the result is negative.
If products have higher enthalpy, the result is positive.
Units of Delta H
Delta H is usually measured in:
- Joules (J)
- Kilojoules (kJ)
Because chemical reactions often involve large energy changes, kilojoules per mole (kJ mol⁻¹) is the most common unit.
For example:
ΔH = −92 kJ mol⁻¹
This means 92 kilojoules of heat are released per mole of reaction.
Types of Delta H in Chemistry
Delta H can describe different kinds of processes. Each type has a specific name.
Enthalpy Change of Reaction
This is the general Delta H for any chemical reaction.
It represents the heat change when reactants turn into products under standard conditions.
Enthalpy Change of Formation
This refers to the heat change when one mole of a compound is formed from its elements in their standard states.
It is often written as:
ΔHf°
These values are widely used in thermochemistry calculations.
Enthalpy Change of Combustion
This describes the heat released when one mole of a substance burns completely in oxygen.
Combustion enthalpy values are always negative because combustion releases heat.
Enthalpy Change of Neutralization
This is the heat change when an acid and a base react to form water.
For strong acids and bases, this value is nearly constant.
Real World Examples of Delta H
Delta H is not just a classroom concept. It plays a role in everyday life and industry.
Examples include:
- Fuel efficiency in engines
- Energy production in power plants
- Heat packs and cold packs
- Food calorie calculations
In each case, Delta H helps explain where energy comes from and where it goes.
Delta H vs Delta G: A Common Comparison
Delta H is often compared with another thermodynamic quantity called Delta G.
Comparison Table
| Delta H | Delta G |
|---|---|
| Measures heat change | Measures free energy change |
| Focuses on enthalpy | Includes entropy effects |
| Indicates heat absorbed or released | Indicates spontaneity |
| Used in thermochemistry | Used in thermodynamics |
Although Delta H is important, it does not alone determine whether a reaction will occur spontaneously.
Common Misconceptions About Delta H
Many learners misunderstand Delta H at first. Clearing these misconceptions helps build confidence.
Delta H Does Not Measure Reaction Speed
tells you how much heat is involved, not how fast a reaction happens.
Delta H Is Not Total Energy
It only measures enthalpy change, not all forms of energy in a system.
A Negative Delta H Does Not Always Mean Spontaneous
A reaction can release heat and still not happen without external input.
Delta H in Laboratory Experiments
In laboratories, Delta H is often measured using calorimetry.
Calorimeters measure temperature changes in surroundings to calculate heat flow.
From these measurements, scientists determine Delta H values accurately.
Symbol Meaning of Delta in Delta H
The Greek letter Delta (Δ) always means change in science.
Therefore:
- ΔH means change in enthalpy
- ΔT means change in temperature
- ΔV means change in volume
Understanding this makes chemical symbols easier to interpret.
Alternate Meanings of Delta H
In chemistry and physics, Delta H almost always refers to enthalpy change.
However, in other fields:
- Delta may represent change in general
- H may represent different variables depending on context
Within chemistry, though, Delta H consistently means enthalpy change.
Frequently Asked Questions
1. What does Delta H stand for in chemistry?
Delta H stands for the change in enthalpy during a process.
2. Is Delta H heat or temperature?
Delta H represents heat energy change, not temperature.
3. Why is Delta H measured at constant pressure?
Most chemical reactions occur at atmospheric pressure, making enthalpy practical to use.
4. Can Delta H be zero?
Yes. If no heat is absorbed or released, Delta H equals zero.
5. What does a negative Delta H mean?
It means heat is released and the reaction is exothermic.
6. What does a positive Delta H mean?
It means heat is absorbed and the reaction is endothermic.
7. Is Delta H the same as internal energy?
No. Delta H includes internal energy plus pressure volume work.
8. How is Delta H measured experimentally?
It is measured using calorimetry and temperature changes.
Conclusion
So, what does Delta H mean in chemistry? It represents the change in enthalpy, or heat energy, during a chemical or physical process at constant pressure.
To recap:
- Delta means change and H means enthalpy
- Delta H shows whether heat is absorbed or released
- Positive values indicate endothermic reactions
- Negative values indicate exothermic reactions
- Delta H is essential for understanding energy changes in chemistry
Once you understand Delta H, thermochemistry becomes far more intuitive and logical.
Discover More Post
What Does Chale Mean in Spanish? | (Definition, Origins …
What Does THX Mean in Text | Definition, Usage & Examples